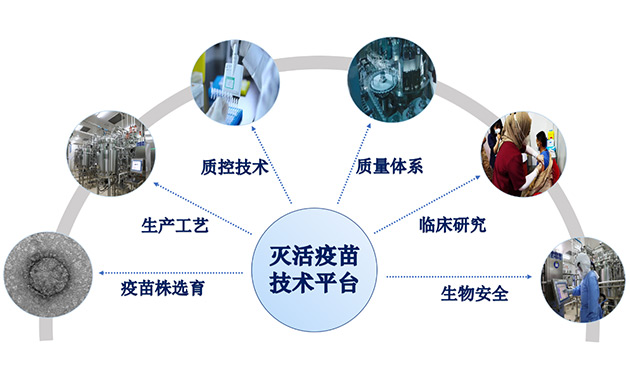
Inactivated vaccine technology platform
BIBP has an international leading technology platform for rapid R&D and industrialization of inactivated vaccines, and has reached the international advanced level in terms of cell substrate establishment, vaccine strain selection and evaluation methods, industrial-grade seed bank system management with international standards, key technologies for large-scale production, development of new quality control technologies, and quality management system.
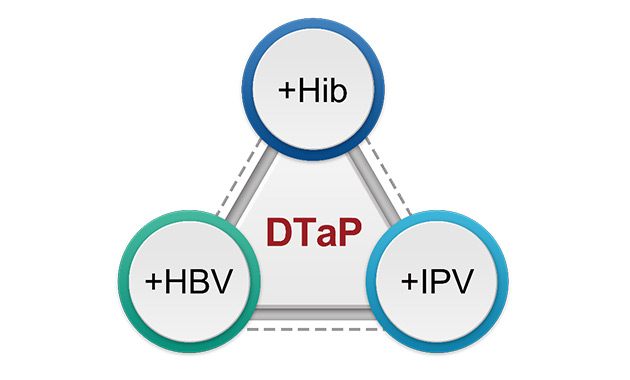
Multivalent vaccine technology platform
BIBP has an international combined multivalent vaccine R&D technology platform, with independent R&D modules in preparation development, adjuvant development, immune effect evaluation, vaccine manufacturing and quality control study. In addition, MMRV vaccine, adsorbed DTaP-poliomyelitis vaccine, and multivalent inactivated COVID-19 vaccine are being developed. The research projects have been supported by National Major Scientific and Technological Special Project for Significant New Drugs Development and Beijing Scientific and Technological Special Project.

Live attenuated vaccine technology platform
In the mid-1950s, the Beijing Institute of Biological Products has carried out technical research and product development of live attenuated vaccines with a long history. We have successfully developed attenuated measles vaccine, rubella vaccine, poliomyelitis vaccine, combined mumps vaccine and other varieties. He has accumulated rich experience in virus breeding and evaluation, sterility control and production management.

Highly pathogenic pathogen vaccine manufacturing platform
BIBP has a high-level biosafety production facility cluster, which is the first in China and the largest in the world, serving as an important base for the development and large-scale production of vaccines against emerging and sudden pandemic infectious diseases in China. BIBP has adopted the internationally advanced GMP concept for drug production, and referred to the EU and WHO standards in terms of critical process parameters, quality attributes and quality standards during operation. Its related hardware standards and management systems have been promoted to other vaccine manufacturers in China.
