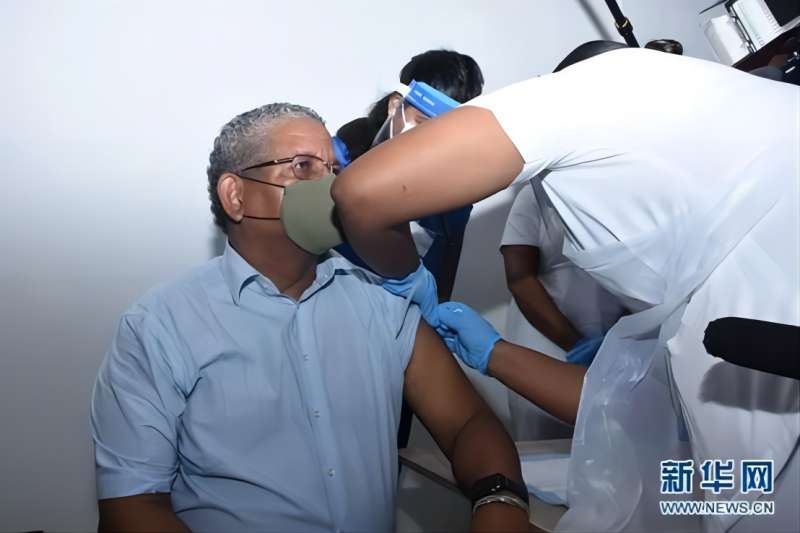
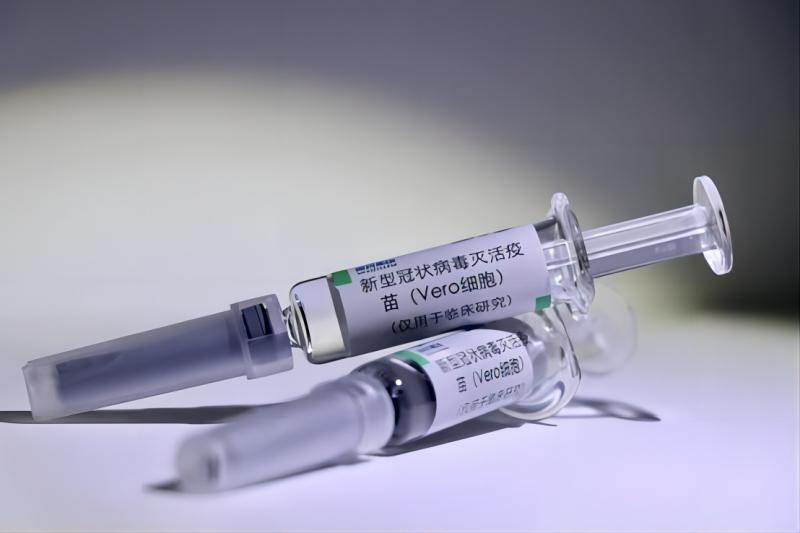
Seychelles Becomes the Fourth Country that Approves BIBP’s COVID-19 Vaccine for Emergency Use
On February 25, 2021, the Ministry of Health (MOH) of Seychelles officially approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (BIBP’s COVID-19 Vaccine) for registration and marketing in Seychelles, making Seychelles the fou...
View More +Hungary — The First EU Member that Approves BIBP’s COVID-19 Vaccine
Inactivated COVID-19 Vaccine (Vero Cell) of Beijing Institute of Biological Products Müller Cecília, Chief Medical Officer of Hungary, announced at a press conference on January 29 that, Hungary’s drug regulator gave use approval to the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., L...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Iraq, Pakistan and Morocco!
Approved for Emergency Use in Iraq The Iraqi National Regulatory Authority has approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (“BIBP’s COVID-19 Vaccine”) for emergency use, as reported by Iraqi media on Ja...
View More +