Fighting against COVID-19
BIBP plays its role wherever there is an epidemic. In the fight against the COVID-19, under the unified deployment and command of Sinopharm, BIBP forged ahead and raced against time to develop the inactivated COVID-19 vaccine, and made a series of world-leading major innovative achievements. After the inactivated COVID-19 vaccine was developed successfully, BIBP has been playing a positive role in guarantee supplies to meet domestic needs, and providing vaccines to countries and international organizations in need as well. So far, BIBP’s COVID-19 vaccine has been approved for emergency use or market access in 119 countries, regions and international organizations worldwide, and has been inoculated by more than 50 heads of state and government.
The first
Chinese COVID-19 vaccine that has been marketed globally
The first
COVID-19 vaccine from non-Western countries that was certified by the WHO
The first
Chinese COVID-19 vaccine that was granted GMP certification by the EU
R&D History
In April, 2020, BIBP’s COVID-19 vaccine took the lead in obtaining the clinical trial approval.
In June, 2020, the world’s first international clinical trial (Phase III) for the inactivated COVID-19 vaccine was officially launched for BIBP’s COVID-19 vaccine.
In July, 2020, BIBP’s COVID-19 vaccine was approved for emergency use (for adult).
In December, 2020, BIBP’s COVID-19 vaccine became the world's first inactivated COVID-19 vaccine to be conditionally approved for marketing.
In April, 2021, BIBP’s COVID-19 vaccine was issued EU GMP compliance certificate by Hungary National Institute of Pharmacy and Nutrition
In May, 2021, BIBP’s COVID-19 vaccine was approved for WHO “Emergency Use Listing (EUL)”, becoming the world's first non-Western country COVID-19 vaccine approved by WHO.
In June, 2021, the first batch of BIBP’s COVID-19 vaccine supplied for WHO COVAX was rolled off the production line.
In July, 2021, BIBP’s COVID-19 vaccine was approved for emergency use in 3 to 17-year-old population.
In August, 2021, BIBP’s COVID-19 vaccine was approved for use in over 100 countries.
In April, 2022, BIBP’s Omicron strain COVID-19 vaccine obtained clinical trial approval.
Workshop
BIBP has three production workshops with a hundred-of-million production scale of COVID-19 Inactivated Vaccine (Vero Cell) , which ensures sufficient vaccine supply and contributes to the humankind common health construction.
58Day
Built the very first P3 production workshop for inactivated COVID-19 vaccine in China in 58 days
90Day
Built the phase II production workshop in 90 days to continuously provide sufficient high-quality COVID-19 vaccines to the world
98Day
Built the world’s largest production workshop for inactivated COVID-19 vaccine in 98 days, and further expand its production capacity




Related News
Glad Tidings! Top 100...
2022-12-30
On December 22, Beijing Enterprise Confederation and Beijing Enterprise Directors Association released the “1+5" lists of 2022 Top 100 Beijing Enterprises, 2022 Top 100 Beijing Manufacturing Enterprises, and 2022 Top 100 Beijing ...
View More+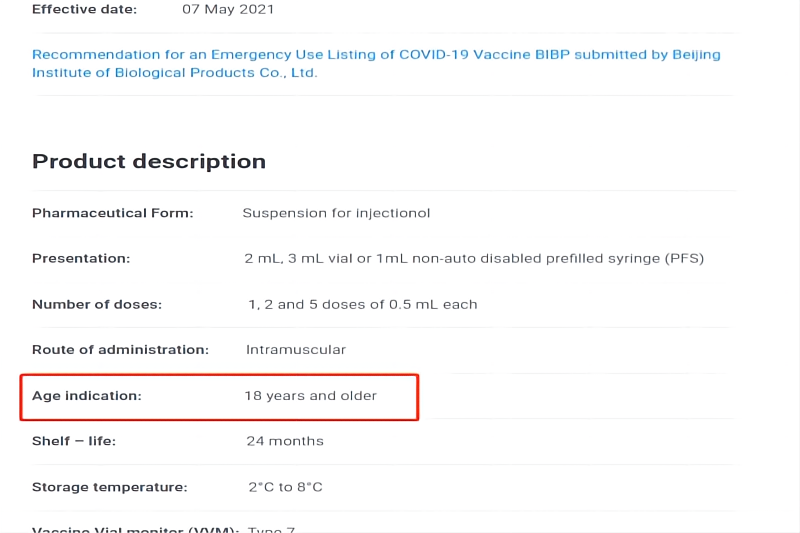
Significant Progress!...
2022-12-03
Recently, WHO has extended the recommended age of use of COVID-19 Vaccine (Vero Cell), Inactivated developed and produced by SINOPHARM CNBG Beijing Institute of Biological Products Co., Ltd. (BIBP) ...
View More+
Glad Tidings! Product...
2022-11-24
Recently, the much-anticipated “2022 Dingge Award—Digital Transformation Pioneer List” was announced and Beijing Institute of Biological Products (BIBP) subordinate to China National Biotec Group (CNBG) of China National Ph...
View More+
Big News!!! BIBP’s In...
2022-04-29
On April 29, theOmicron COVID-19 Vaccine (Vero Cell), Inactivated developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sino...
View More+